hicAggregateContacts¶
Background¶
hicAggregateContacts is a tool that allows plotting of aggregated Hi-C sub-matrices of a specified list of positions. Positions of interest can for example be binding sites of a specific protein that were determined by ChIP-seq or genetic elements as transcription start sites of active genes.
Description¶
Takes a list of positions in the Hi-C matrix and makes a pooled image.
usage: hicAggregateContacts --matrix MATRIX --outFileName OUTFILENAME --BED
BED --range RANGE [--BED2 BED2]
[--numberOfBins NUMBEROFBINS]
[--transform {total-counts,z-score,obs/exp,none}]
[--avgType {mean,median}] [--help] [--version]
[--outFilePrefixMatrix OUTFILEPREFIXMATRIX]
[--outFileContactPairs OUTFILECONTACTPAIRS]
[--diagnosticHeatmapFile DIAGNOSTICHEATMAPFILE]
[--kmeans KMEANS] [--hclust HCLUST]
[--howToCluster {full,center,diagonal}]
[--chromosomes CHROMOSOMES [CHROMOSOMES ...]]
[--colorMap COLORMAP] [--plotType {2d,3d}]
[--vMin VMIN] [--vMax VMAX] [--dpi DPI]
Required arguments¶
- --matrix, -m
Path of the Hi-C matrix to plot.
- --outFileName, -out
File name to save the image.
- --BED
Interactions between regions in this BED file are plotted.
- --range
Range of contacts that will be considered for plotting the aggregate contacts in bp with the format low_range:high_range for example 1000000:20000000. The range should start at contacts larger than TAD size to reduce background interactions.
Optional arguments¶
- --BED2
Optional second BED file. Interactions between regions in first and second BED file are plotted.
- --numberOfBins
Number of bins to include in the submatrix. The bed regions will be centered between half number of bins and the other half number of bins.
Default: 51
- --transform
Possible choices: total-counts, z-score, obs/exp, none
Type of transformation for the matrix. The options are “none”, “total-counts”, “z-score” and “obs/exp”. If total counts are selected, the sub-matrix values are divided by the total counts for normalization. If z-score or obs/exp are selected, the Hi-C matrix is converted into a z-score or observed / expected matrix.
Default: “none”
- --avgType
Possible choices: mean, median
Type of average used in the output matrix. Options are mean and median. Default is median.
Default: “median”
- --version
show program’s version number and exit
- --dpi
Optional parameter: Resolution for the image in case theouput is a raster graphics image (e.g png, jpg).
Default: 300
Output options¶
- --outFilePrefixMatrix
If this option is set, the values underlying the output matrix will be saved to tab-delimited tables (one per chromosome) using the indicated prefix, for example TSS_to_TSS_chrX.tab. If clustering is performed, the values are saved including the cluster_id in the file TSS_to_TSS_chrX_cluster_1.tab.
- --outFileContactPairs
Output file prefix. If this option is set, the position of the contact positions are saved as (chrom1, start1, end1, chrom2, start2, end2) where chrom_n, start_n, end_n correspond to the pair of positions used to compute the submatrix. The data is saved per chromosome and per cluster separately (one file each). The format is {prefix}_{chrom}_{cluster_id}.tab. If no clusters were computed, then only one file per chromosome is produced.
- --diagnosticHeatmapFile
If given, a heatmap (per chromosome) is saved. Each row in the heatmap contains thediagonal of each of the submatrices centered on the bed file. This file is useful to get an idea of the values that are used for the aggregate matrix and to determine the fraction of sub-matrices that are aggregated that may have an enrichment at the center.
Clustering options¶
- --kmeans
Number of clusters to compute. When this option is set, the submatrices are split into clusters (per chromosome) using the k-means algorithm.
- --hclust
Number of clusters to compute (per chromosome). When this option is set, the matrix is split into clusters using the hierarchical clustering algorithm, using “ward linkage”. –hclust could be very slow if you have >1000 submatrices per chromosome. In those cases, you might prefer –kmeans
- --howToCluster
Possible choices: full, center, diagonal
Options are “full”, “center” and “diagonal”. The full clustering is the default and takes all values of each submatrix for clustering. “center”, takes only a square of length 3x3 from each submatrix and uses only this values for clustering. With the “diagonal” option the clustering is only carried out based on the submatrix diagonal (representing values at the same distance to each other.)
Default: “full”
Plotting options¶
- --chromosomes, -C
List of chromosomes to plot.
- --colorMap
Color map to use for the heatmap. Available values can be seen here: http://matplotlib.org/examples/color/colormaps_reference.html
Default: “RdYlBu_r”
- --plotType
Possible choices: 2d, 3d
Plot type.
Default: “2d”
- --vMin
Minimum value of the plotted score.
- --vMax
Maximum value of the plotted score.
Usage example¶
Below, you can find an example of an aggregate Hi-C matrix obtained from Drosophila melanogaster Hi-C data. The interactions are plotted at binding sites of a protein that were determined by ChIP-seq. We plot sub-matrices of 30 bins (1.5 kb bin size, 45 kb in total). The regions specified in the BED file will be centered between half number of bins and the other half number of bins.The considered range is 300-1000 kb. The range should be adjusted and only contain contacts larger than TAD size to reduce background interactions.
$ hicAggregateContacts --matrix Dmel.h5 --BED ChIP-seq-peaks.bed \
--outFileName Dmel_aggregate_Contacts --vMin 0.8 --vMax 2.2 \
--range 300000:1000000 --numberOfBins 30 --chromosomes X \
--avgType mean --transform obs/exp
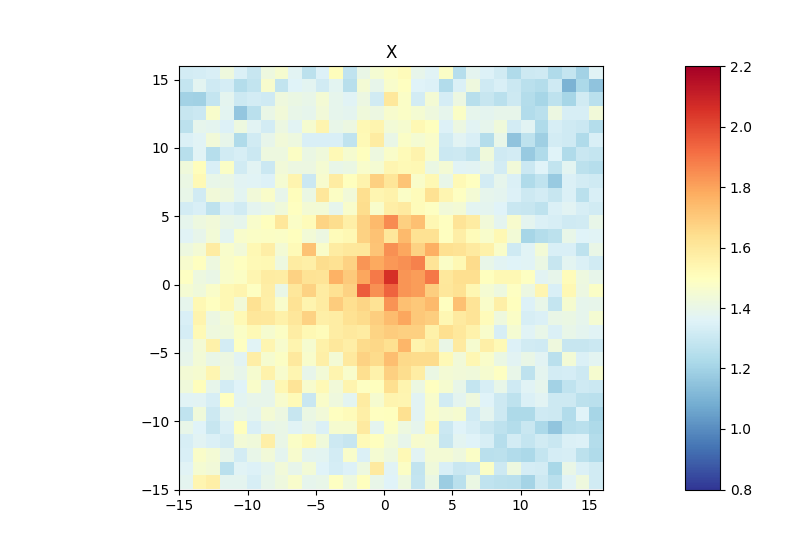
This example was calculated using mean interactions of an observed vs expected transformed Hi-C matrix. Additional options for the matrix transformation are total-counts or z-score. Aggregate contacts can be plotted in 2D or 3D.